The perfect peak shape: Five solutions to peak tailing problems
For chromatographers, there is no sight more beautiful to behold than perfectly symmetrical peak shapes! But how do we even measure peak shape symmetry. And do perfect peaks really exist? Certainly not when you have peak tailing problems! Read on to find out more about perfectly symmetrical chromatography peaks and five ways to achieve them by avoiding peak tailing.
My wife and I were out for a stroll the other day when she noticed a young woman wearing a very interesting skirt made of different fabrics that were asymmetrically cut. My wife commented that she liked the skirt but noted that her mother would have absolutely hated it. Apparently, it was very important to have perfectly cut, straight, symmetrical skirts back in the days. She argued that symmetry is just more appealing to humans.
Well, beauty is in the eye of the beholder. I don’t care much for symmetrical skirts, symmetrical faces or symmetrical flowers. But as a scientist, I do like my symmetry. Come on, who didn’t get excited this week about the possible detection of an axion, the lightest predicted supersymmetry particle, underneath an Italian mountain last month?
And as a chromatographer, oh boy. There is nothing like those perfectly symmetrical peaks in a chromatogram to get me going!
We all know perfect symmetrical peak shapes obtain the best results, but sadly, reality hardly ever results in perfect situations. How about we go on to explore peak shapes in a bit more detail?
I suggest we start off by reviewing how the symmetry of a peak is measured. There are two main ways to do this.
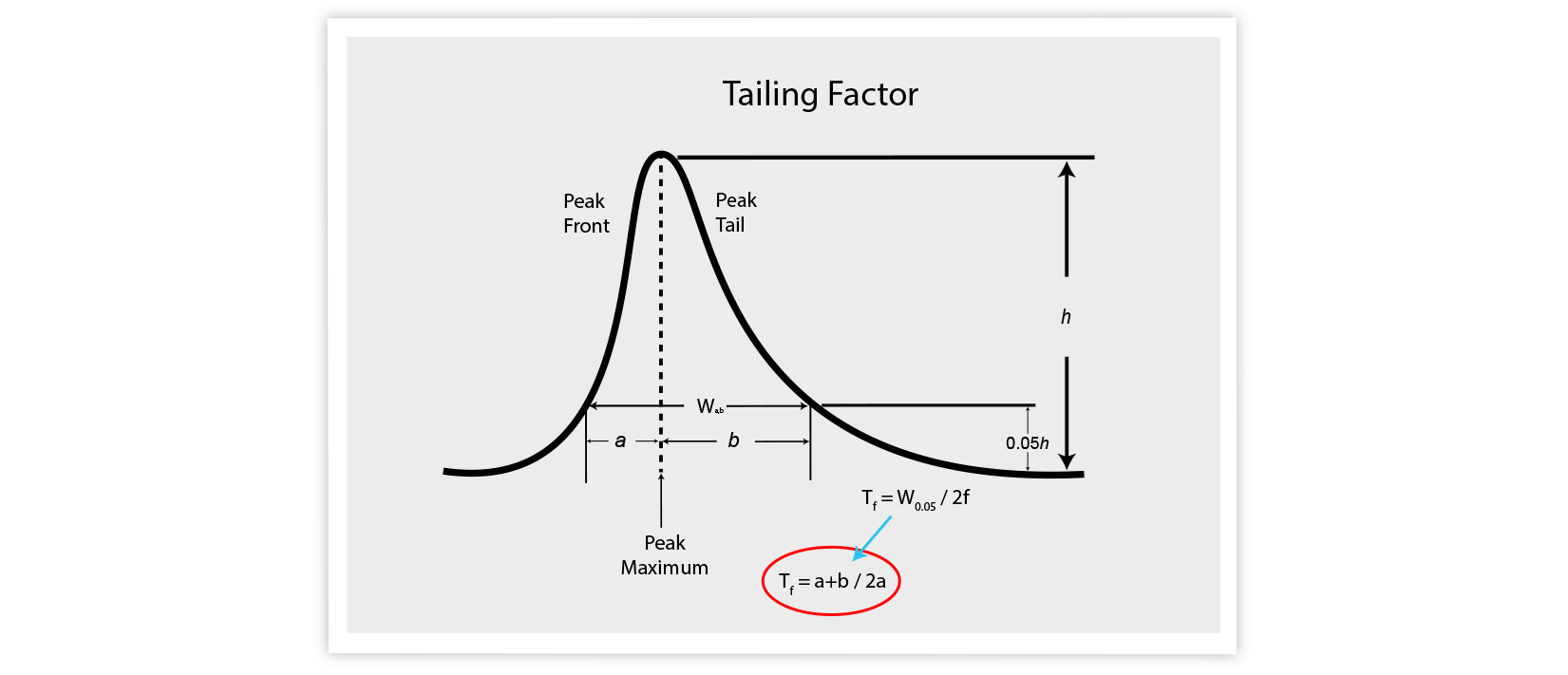
The first method is based on the tailing factor ( Tf ). Here, you measure the peak width at 5% of the peak height above the baseline. This approach is often used in the pharmaceutical world.
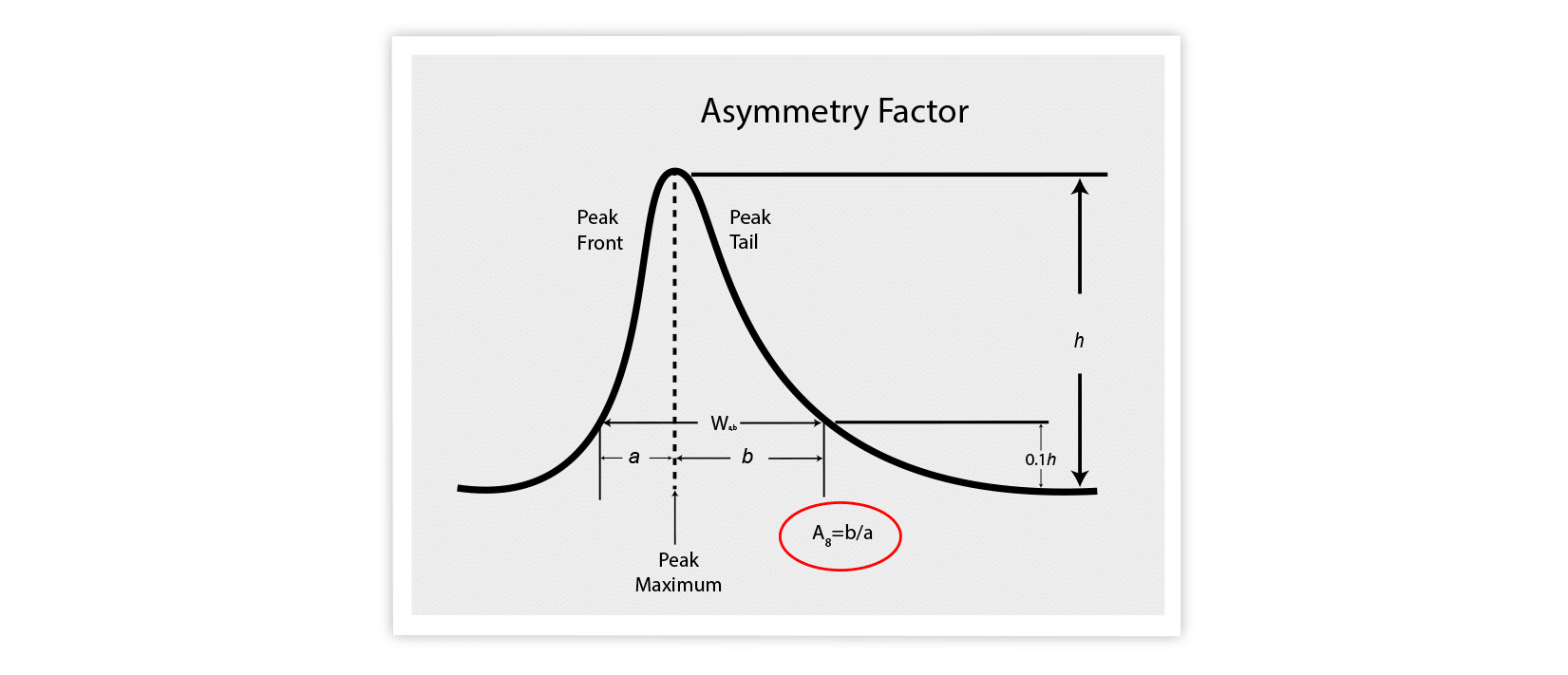
The other method relies on the asymmetry factor ( As ). Here you measure at 10% of peak height above the baseline.
A peak with:
Tf or As = 1 is perfectly symmetrical.
So now we know how to measure peak symmetry. But as I already mentioned, like people, skirts and flowers, chromatography peaks come in all shapes and sizes. Unfortunately, in some cases, this can be rather problematic. I’d like to go on to discuss one frequently encountered phenomenon with you here, mainly peak tailing!
Back to our chromatogram, peaks with:
Tf or As >1 are tailing.
What causes peak tailing?
Well, there are various possible causes of this peak shape.
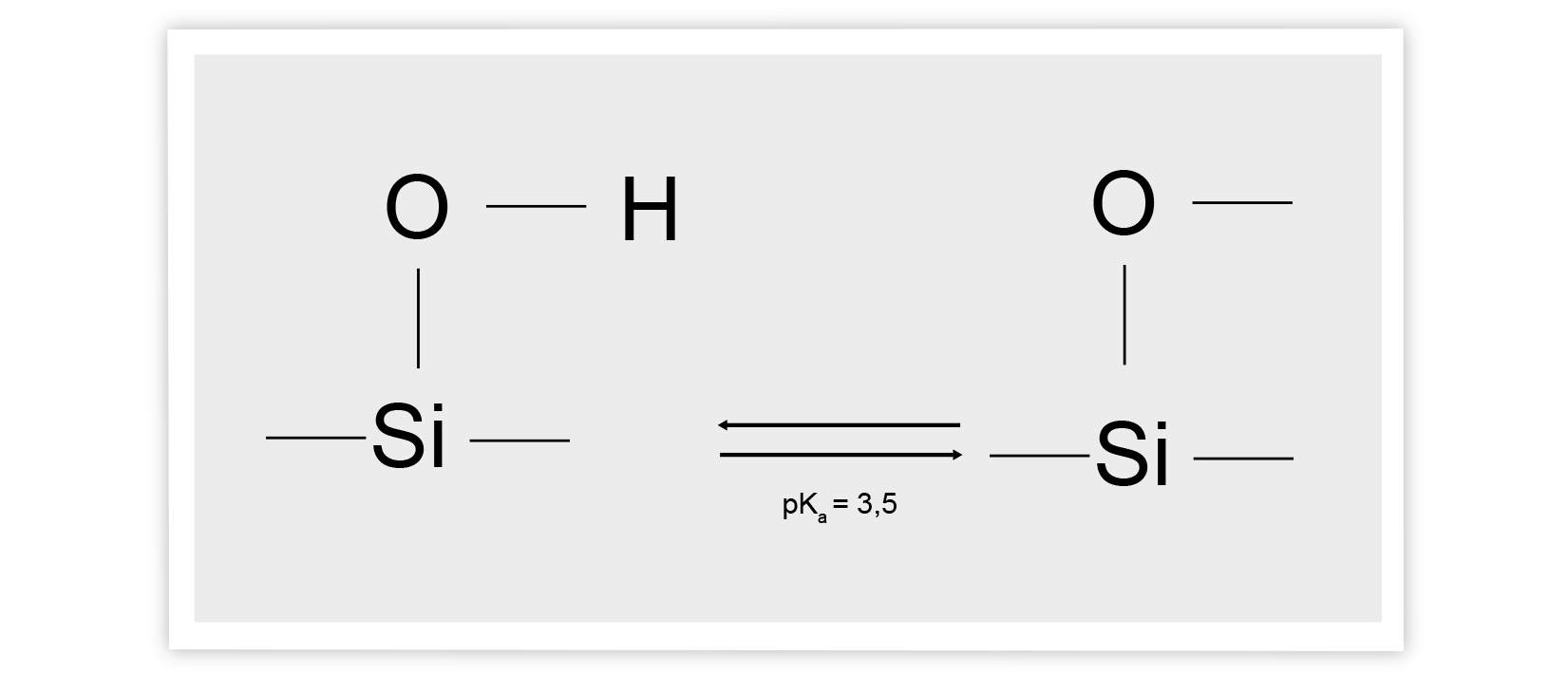
Cause 1: Firstly, tailing can occur when secondary interactions take place. As a result, not all molecules travel through the column at the same speed and this causing tailing at the peak.
Possible Solutions: To remedy this, you could try to lower the pH of the liquids so that silanol ionization is suppressed (pH 3). Reducing the pH keeps the silanols in protonated form, where interaction with polar compounds is minimized. This, in turn, has a positive effect on your peak shape.
Alternatively, you could use packing material with better end-capping to reduce the surface activity. Lastly, you could try adding buffers to the mobile phase running through your chromatography system. For purification purposes, these buffers should be volatile, so that they can easily evaporate with the mobile phase after sample collection.
Cause 2: Tailing can occur due to packing bed deformation. The creation of a void at the inlet of the column or the presence of channels in the packing bed can initially lead to peak tailing. Eventually, this phenomenon could lead to peak splitting, another problematic shape I’ll discuss in a future post.
Additionally, collection of particles at the inlet frit can lead to peak tailing.
Possible solutions: Apply extreme care when self-packing columns or purchase pre-packed consumables if possible. Filtration of the samples prior to introduction on the column can help prevent peak tailing resulting from collection of particles at the inlet frit.
Cause 3: Tailing can occur due to column overload
Possible solutions: You could try using a bigger column or decreasing the sample amounts introduced to the column to eliminate the problem.
Cause 4: Sometimes, the presence of excessive extra column dead volume can result in peak tailing. This problem usually effects the peak shape of the early eluting peaks. Later eluting compounds will be more influenced by the packing material itself as these reside longer in the material.
Possible solutions: Again, exercise extra caution when pre-packing your column.
Cause 5: The purity of the packing material can also influence peak shape. Contaminants, such as trace metals, in the material can enhance secondary interactions, which will lead to peak tailing.
Possible solutions: I would recommend using the purest packing material possible to help fix this problem.
So there you have it. Five causes of peak tailing and five remedies to help you leave this problematic peak shape behind. Unfortunately, I also have some bad news. There are more nasty peak shapes that can occur during your purifications. But I would like to leave you with some good news. I will offer you some ideas on how to eliminate other asymmetrical peaks in a future post. And I promise, I will not keep you waiting long.
Till next time,

WANT TO STAY IN TOUCH?
Click on the button and receive the latest posts directly in your messenger!
Related Posts
16th November 2023
Warm and Colorful Goodbye from Bart
After 136 posts filled with knowledge, Bart announced his retirement. Read here his goodbye message. →
9th November 2023
Go Big or Go Home: Scaling-up Spray Drying Processes from Laboratory to Industrial Scale
Bart explains the differences between performing spray drying on a laboratory and industrial scale. →
24th October 2023
Don’t be a Drip: Perform Chromatography in a Flash
Bart describes the processes of open column and flash chromatography and explains the critical factors that need to be considered when switching approach. →
5th October 2023
Protein Preservation: Critical Factors Affecting Freeze Drying Formulations
Bart compares freeze drying formulas with soil preparation to explain how best to preserve and freeze dry proteins and peptides. →
21st September 2023
In Pursuit of Purity: Chiral Column Screening and the Separation of Ketaprofen using SFC
Bart explains the importance of column screening in SFC, using the example of separating ketoprofen. →
6th September 2023
Understanding Energy Balance to Optimize Lab Efficiency: Rotary Evaporation
Bart explains how the Laws of Thermodynamics influence energy balance in the laboratory. →
23rd August 2023
Keeping your Cool: Essential Tips for Freeze Dryer Maintenance
Bart gives some useful tips for how to maintain freeze drying equipment to ensure its longevity and guarantee optimal performance. →
9th August 2023
A Tale of Two Techniques: How to Choose between prep HPLC and prep SFC
Bart explains how to choose between prep HPLC and prep SFC. →
26th July 2023
The Explosive Truth: Unpacking the importance of ATEX certification.
Bart explains the ATEX directives and how EX-proof instruments ensure safety in high-risk environments. →
12th July 2023
A Breath of Fresh Air: Formulating Inhalable Drugs through Spray Drying
Bart explains how inhalable drugs work and how to create them using spray drying. →
29th June 2023
As Easy as ABC: How to Use the Van Deemter Equation to Optimize Your Chromatography
Bart explains the Van Deemter Equation and explains how it is used to optimize different chromatography methods, such as, prep HPLC and prep SFC. →
14th June 2023
The Evolution of Evaporation: From Ancient Alchemy to Modern Science
In this blog, I would like to talk about the history and future of rotary evaporation, a process I have had tremendous experience with over the years. →
31st May 2023
Chilling Out! How to perfect the freeze drying process
The importance of spray drying , I would like to take the time to focus on a different method used to remove moisture from a product. →
17th May 2023
Chiral Compound Confusion: Bart explains how SFC is used to purify enantiomers.
I would like to focus specifically on chiral compounds, also known as enantiomers. →
3rd May 2023
Getting Hands-on with SFC: Bart explores the application possibilities of supercritical fluid chromatography
Explaining what applications are possible with SFC →
19th April 2023
Maintenance Matters! The importance of keeping your equipment in tip-top shape
The power of prevention is of particular importance when it comes to the maintenance of laboratory equipment. →
5th April 2023
The Never Contented! How spray drying enables batteries to keep on keeping on.
Bart explains how spray drying technology is used to improve battery performance. →
22nd March 2023
Stay cool and protect your analytes: Maceration vs. continuous cold extraction on a rotavapor
In this post, I shall compare two methods for performing extractions on heat-sensitive samples. →
10th March 2023
Shining a light on automated evaporation solutions
Bart explains how the latest automated features on rotary evaporators makes the sample preparation process of solid loading a breeze. →
2nd March 2023
Let’s get critical! SFC vs. HPLC
Bart explains the history of SFC and how it has developed over the years. SFC is then compared with HPLC. →
8th February 2023
Celebrating chromatography for the blog’s 5th blooming birthday
Bart celebrates the blogs 5th year by explaining how chromatography was invented by Mikhail Tsvet. A simple experiment is described that explains the key concepts. →
11th January 2023
What’s coming up in 2023
The New Year always provides an opportunity to look forward and to reflect. As I have embraced retirement, I have found more time for reflection. →
22nd December 2022
Develop flash and prep HPLC applications to purify a range of compounds
Bart explains the versatility of Flash and Prep HPLC applications to ensure the successful purification. →
5th December 2022
Leaks giving you the freaks? How to perform a leak test on your rotary evaporator
Bart Explains how to find and prevent leaks in rotary evaporators… →
17th November 2022
Flavor of the day: How to spray dry flavors and fragrances part 2!
Bart describes spray drying parameters and particle characteristics when encapsulating flavors and fragrances →
2nd November 2022
A fat lot of tips on how to purify lipids
Bart offers advice on how to purify lipids, including phase selection, sample loading and detection methods →
19th October 2022
A whiff of microencapsulation of flavour and fragrances by spray drying
Bart goes over the benefits of microencapsulation, carrier materials and factors affecting emulsification →
5th October 2022
Pump it up: clean vacuum pump in six easy steps
Bart explains when you should clean your vacuum pump and how to most efficiently get the job done →
21st September 2022
Solving the chromatography solvent problem
Bart examines mobile phases used in normal and reversed-phase chromatography →
9th September 2022
The role of spray drying in gene therapy
Bart offers spray drying parameters from literature on spray drying nucleic acids for delivery approaches in gene therapy →
2nd September 2022
Taking the heat off cleaning your heating bath
Bart goes through the 12 simple steps you need to clean the heating bath of your rotary evaporator →
10th August 2022
How to spray dry nucleic acids
Bart explains why and how you should spray dry nucleic acids for gene therapy →
27th July 2022
Your supplement on vitamin purification
Bart discusses how to adapt chromatography methods to vitamin applications →
13th July 2022
How to clean the glassware of your rotary evaporator
Bart highlights seven steps for cleaning the inner and outer glassware parts of a rotary evaporator →
29th June 2022
This is what a next-generation spray dryer looks like
Bart introduces a new spray drying system and offers six points on why the instrument is a must for laboratory spray drying →
20th June 2022
Why HILIC is what your polar compounds need for purification
Bart explains how HILIC works and why it is a suitable method for separating polar substances →
1st June 2022
Four rotary evaporation tricks to keep your safety standards high!
Bart highlights the importance of plastic-coated glassware, secondary condensers, splash shields and level sensors for safety during rotary evaporation →
18th May 2022
Episode 100: An investigation into freeze-drying and spray-drying effects on peptides
Bart reviews a paper on effects of freeze drying vs. spray drying on bioactives →
4th May 2022
How to use your rotary evaporator for freeze-drying sample preparation
Bart guides you through using a Dewar accessory on a rotary evaporator for lyophilization sample prep →
21st April 2022
Don’t mess with my flow! How to solve three common solvent flow problems in chromatography
Bart helps diagnose and cure solvent flow problems →
6th April 2022
Watering your knowledge on spray drying and freeze drying
Bart offers a general overview and comparison of spray drying and freeze drying →
23rd March 2022
Flash chromatography vs prep HPLC: you want speed or precision?
Bart compares open column chromatography, flash chromatography and prep HPLC →
9th March 2022
Five glassware accessories that those doing rotary evaporation swear by
Bart discusses the importance of five glassware accessories in rotary evaporation →
23rd February 2022
Are compounds in your sample ghosting your chromatography detection method?
Bart gives an overview of theory, benefits and limitations of five common detection methods in chromatography →
14th February 2022
Super special edition: Interview with Bart for blog’s 4th birthday!
Bart participates in a personal Q&A offering insights into his life and his science interests →
26th January 2022
Who is your favorite member of the rotary evaporation family?
Bart matches industrial, parallel and lab evaporators to their most suitable applications →
12th January 2022
What’s written in the stars for rotary evaporation, chromatography and spray drying?
Bart makes his 2022 predictions for chemical and pharma R&D labs →
17th December 2021
All I want for Christmas is some chemistry webinars!
Bart gives you lots of holiday viewing with webinars on chromatography, proteins, natural products and more →
1st December 2021
What’s the BIG deal with industrial evaporation?
Bart discusses differences and how to upscale from lab to industrial evaporation →
17th November 2021
Three chromatography system parts and why they are important
Bart focuses on use and importance of fittings, hoses & tubes and ports in chromatography →
3rd November 2021
How to make your protein formulation last and last and last
Bart offers tips on using spray drying and freeze drying for protein formulations →
20th October 2021
Are you team “spray dry” or “freeze dry” when you formulate proteins and peptides?
Bart compares spray drying and freeze drying for protein applications →
6th October 2021
Five essential steps in the drug discovery and development process
Bart discusses the methods for extraction, concentration, purification and formulation of APIs →
22nd September 2021
Two reasons why prep HPLC users love analytical chromatography
Bart explains how to use analytical chromatography to develop an optimized prep HPLC method →
8th September 2021
Why you should avoid grease in your rotavapor with a PTFE Stopcock
Bart describes the dangers of grease for your rotary evaporation and how to use a PTFE stopcock to solve this challenge →
3rd September 2021
This method of concentrating natural products is really a force of nature
Bart explains how to use rotary evaporation to concentrate natural ingredients →
11th August 2021
Tickling those taste buds: Rotary evaporation for flavour creation
Bart offers his chef's kiss on how to use rotary evaporation to create flavours for mixologists and chefs →
14th July 2021
How to freeze dry PCR diagnostic kits for COVID-19
Bart details the five steps needed for the lyophilization of PCR diagnostic kits for various diseases →
30th June 2021
Concentrate on this: Tips for concentration of proteins and peptides
Bart offers tips on how to optimize rotary evaporation and freeze drying for protein concentration →
17th June 2021
Is your evaporation instrument the perfect match to your application?
Bart looks at different applications of rotary evaporation and which evaporation system is better suited to them →
2nd June 2021
Rotary evaporation vs freeze drying in protein concentration
Bart lists factors that help determine if rotary evaporation or freeze drying is more suitable for your protein concentration →
31st May 2021
Let’s match a vapor duct to your rotary evaporation application
Bart discusses three applications of rotary evaporation and what vapor duct is most suitable for each one →
5th May 2021
Tips on equilibration and storage of flash cartridges and prep HPLC columns
Bart offers insights into how to properly equilibrate and store your flash cartrdiges and prep HPLC columns →
21st April 2021
Seven indicators your rotary evaporation glassware is of good glass quality
Bart explains the telltale signs of good quality glassware used in rotary evaporation →
7th April 2021
Purification of natural compounds can come this naturally to you. Here is how.
Bart gives advice on how to develop a chromatography method for purification of natural products →
24th March 2021
Not just another SOP story: How to program a rotary evaporator method
Bart shows how to divide an SOP into multiple steps and parameters that can be entered into the interface of a rotary evaporator →
10th March 2021
How to find the perfect stationary phase for your protein purification
Bart discusses what types of chromatography and stationary phases are well suited to protein purification applications →
1st March 2021
Three parameters you need to boost your distillation rate
Bart shows how to improve distillation rates through optimal heating bath temperature, condenser loading and flask size →
15th February 2021
Happy birthday to me: The blog turns three and it’s all about the accessories!
Bart celebrates the blog's birthday with resources on accessories in chromatography and rotary evaporation →
27th January 2021
How to efficiently maintain a flash chromatography system
Bart details how to take care of the pump, detector and fraction collector of a chromatography system →
13th January 2021
Your 2021 chromatography and rotary evaporation crystal ball
Bart offers his outlook on how the chromatography and rotary evaporation markets will develop in 2021 →
23rd December 2020
Should I stay or should I go
Bart announces his retirement, but promises to continue writing for the blog →
18th December 2020
Let’s wrap up this unusual year with an unusual post
Bart says goodbye to 2020 with four chromatography and rotary evaporation videos for faithful blog readers to enjoy over the holidays →
25th November 2020
7 highly important factors for cannabinoid isolation
Bart lists seven factors that are important for successful of separation of cannabinoids using chromatography →
4th November 2020
Foam is foe in rotary evaporation. Here is how to prevent it.
Bart discusses why foam formation is dangerous and how to prevent foam in your evaporation flask →
21st October 2020
Slurry packing of chromatography columns can be just as fun as it sounds
Bart explains the slurry packing process for chromatography columns →
8th October 2020
Condenser loading should not be at 100% and here is why.
Bart discusses optimal condenser loading and why it is important →
24th September 2020
Dry packing of a chromatography column in three steps
Bart explains how to dry fill a chromatography column →
10th September 2020
Why 15 is the magic number for your recirculating chiller
Bart shows how to calculate cooling capacities and discloses how to set up your recirculating chiller to achieve sufficient condensation →
26th August 2020
How RFID technology lifeguards your chromatography process
Bart explains how and why RFID tags are used on chromatography cartridges and racks →
12th August 2020
Sizing up the importance of flask size in rotary evaporation
Bart discusses theory and experimental data on how flask size affects evaporation performance →
29th July 2020
Are peak splitting and peak fronting effects keeping you up at night?
Bart talks about peak fronting and peak splitting and how to eliminate these effects in chromatography →
16th July 2020
Why you should give the glass thickness of your evaporation flask a second thought
Bart discusses how the glass thickness of a flask affects evaporation performance and safety →
17th June 2020
Five ways to optimize the pressure values of your rotary evaporation
Bart offers five tips for finding the ideal pressure settings for rotary evaporation of a solvent →
3rd June 2020
What’s hidden behind your cosmetics labels? Chromatography!
Bart highlights the role of chromatography in ingredient discovery for development of cosmetics →
20th May 2020
How the delta 20 rule can make all the difference in your rotary evaporation
Bart describes the golden delta 20 rule for finding optimal temperature values for laboratory evaporation →
6th May 2020
The joint relationship between chromatography and cannabis
Bart talks about the purification of cannabinoids like CBD from cannabis using flash chromatography and solvent recycling →
22nd April 2020
Five cases where the bigger the condenser, the better
Bart explores the relationship between condenser size and distillation rate with specific examples and experimental data →
8th April 2020
All about ELS detectors – part 002
Bart discloses valuable information on ELS detectors in his sequel post on the topic of ELSD →
25th March 2020
Eco-friendly rotary evaporation is possible and here is the evidence
Bart shares energy savings data from using eco-friendly features on a rotary evaporator →
11th March 2020
How to solve two challenges of UV detectors in chromatography
Bart gives advice on how to overcome two common challenges of UV detection in chromatography →
26th February 2020
Is your rotary evaporator well configured?
Bart jumps into rotary evaporation by discussing current applications and how to configure your own rotary evaporator →
14th February 2020
The blog turns two, but you get all the presents
Bart's Blog turns two, offers chromatography presents, introduces rotary evaporation as a regular topic →
29th January 2020
How to separate very polar substances without destroying your packing material
Bart shares how to separate very polar compounds using a stationary phase ideal for this purpose →
15th January 2020
What does liquid chromatography have in its 2020 fortune cookie?
Bart shares his predictions on where liquid chromatography is heading in 2020 →
19th December 2019
A look back at your favorite chromatography and freeze-drying posts from 2019
Year-in-review: Bart offers a summary of the most read freeze-drying and chromatography posts in 2019 →
29th November 2019
Why the retention factor needs to be kept in check
Bart offers the ideal retention factor range for achieving good resolution in chromatography while keeping running times and costs low →
14th November 2019
Why a bit of moisture in freeze-dried products can be a good thing
Bart discusses the danger of overdrying during the freeze-drying process and how moisture mapping can help achieve optimal moisture content in products →
23rd October 2019
Does anyone in chromatography give a load about the sample load?
Bart talks about sample overload in chromatography and how it affects plate height, column efficiency and resolution →
9th October 2019
A load of tips on how to prevent condenser overload
Bart describes how freeze-driyng rates, condenser ice capacity and other parameters can be used to prevent condenser overload →
25th September 2019
Five good reasons to attend my live Q&A chromatography webinar
Bart invites readers of his blog to join a live Q&A chromatography webinar where he personally answers their purification questions →
18th September 2019
Simple ways to take the edge off the edge vial effect in freeze drying
Bart draws attention to the edge vial effect and how to reduce its negative impact during the freeze-drying process →
11th September 2019
How to master chromatography in two days
Bart invites users to an advanced user chromatography training →
28th August 2019
The poster that could put Bart’s Blog out of a job
Bart offers a sneak peak into a free chromatography poster with the basic principles needed to perform perfect flash chromatography and prep HPLC →
22nd August 2019
What happens if you run your freeze drying process under direct sunlight
Bart experiments with the effect of direct sunlight on freeze drying performance and draws attention to how the environment can influence lyophilization →
31st July 2019
We need to talk about flow rate and column efficiency in chromatography
Bart discusses how an optimized flow rate helps improve the column efficiency of flash and prep HPLC chromatography →
17th July 2019
How to find a suitable pressure endpoint criterion for freeze drying
Bart discusses comparative pressure measurement and how to use it for endpoint determination of freeze-drying steps →
3rd July 2019
Three major ways to influence column efficiency in your favor
Bart explains how particle size, column packing and pore size affect column efficiency and resolution in chromatography →
27th June 2019
How to use temperature to assess if primary freeze drying is finished
Bart describes how temperature could be used for endpoint determination of the primary drying step in lyophilization →
5th June 2019
Why you should care about column efficiency
Bart explains how column efficiency, or the number of theoretical plates, affect resolution in chromatography →
22nd May 2019
Why pressure gradients are the main drivers in improving sublimation rates
Bart discusses why a good pressure gradient is the most efficient way to improve freeze-drying rates →
8th May 2019
Putting safety first during the chromatography process
Bart discusses how to maximize the safety of user, sample and the surroundings during the chromatography process →
18th April 2019
A pretty hot way to speed up the freeze-drying process
Bart offers theory and experimental data on how heated shelves can speed up the freeze drying process →
10th April 2019
Two essential aids for proper column maintenance
Bart discusses the role of pre-guards, column guards and RFID technology in proper cartridge and column maintenance →
27th March 2019
Shall we use shell freezing to speed up manifold freeze drying?
Bart discusses the benefits of shell freezing over bulk freezing in speeding up the manifold freeze drying process →
13th March 2019
Beware: Protecting yourself and the environment against chromatography hazards
Safe, green chromatography? Yes, you can! →
20th February 2019
How to effectively use organic solvents in lyophilization
Bart offers advice on how to incorporate challenging organic solvents into your laboratory freeze drying process →
14th February 2019
The blog gets older, falls in love and branches out
Bart's blog celebrates its first birthday by welcoming a new guest to the party. Lyophilization will now get regular coverage on the blog! →
7th February 2019
Three chromatography problems the new Pure system solves
There are challenges in chromatography when it comes to user, sample and environmental safety. See how the Pure chromatography system sets out to solve them →
10th January 2019
Four Fab Lab New Year’s Resolutions to live by in 2019
The chromatography team at Büchi lists their chromatography resolutions for 2019 and how they plan to remain funny, innovative, safe and green →
18th December 2018
It’s time we separate from 2018 and flash in a brilliant 2019
Büchi's chromatography team gets ready to bring in the new year by talking about their favorite moments of 2018 and their plans for 2019 →
10th December 2018
How to optimize your mobile phase to improve selectivity and resolution in chromatography
Discover methods for choosing the most suitable solvent to achieve better selectivity and resolution in your separations→
10th December 2018
How to choose a stationary phase, optimize selectivity and get better resolution in chromatography
See the effects of selectivity on separation quality and get some great tips on how to optimize resolution by finding the ideal stationary phase for your application→
29th November 2018
The fastest, easiest and smartest way to select a flash cartridge
Discover a brand new digital tool for flash cartridge selection. Read how the FlashPure Scout app can provide you with tips on finding the ideal cartridge, as well as technical information for method optimization→
30th October 2018
All you need to know about using TLC data for flash method optimization
Convert your TLC results into optimized separations and learn about how certain software can convert TLC data into better purifications →
18th September 2018
Clever ways to speed up laboratory evaporation
This post is a giant turbo button for your laboratory evaporation performance. Read on to gain fresh ideas on how to make your evaporation process faster →
30th August 2018
How to quicken laboratory freeze drying procedures
The freeze drying process in the lab can be fun, but as with all common chemistry methods, the sooner it is done, the better. Learn how to quicken it and download the poster →
3rd August 2018
Top tips on how to save time in chromatography
Get some great suggestions on how you can speed up your chromatography runs and save valuable time →
18th July 2018
The most important factors to consider when selecting a flash chromatography cartridge
See what important parameters you should consider when choosing a flash cartridge and get insights into how to simplify the selection process →
5th July 2018
How to overcome limitations in flash chromatography with an ELSD
Find out how ELSD functions and what benefits this detector can bring to your chromatographic experiments compared to UV detectors alone →
22nd June 2018
World Cup Fever: How to Score Purer Compounds by Improving Resolution in Chromatography
Soccer fans or not, all chemists want to give impurities a red card. Read today how to improve resolution and score purer compounds →
3rd April 2018
How to do sample introduction in Flash Chromatography?
When introducing a sample in flash chromatography, are you going for Wet or Liquid Loading? Learn about the two sample introduction techniques and how they influence the sample loss during the purification process →
7th February 2018
Warm and Colorful Welcome from Bart
Hello there and welcome on the blog!
This place is for all colorful researchers out there who are looking for their next adventure in science. Let's introduce ourselves →
31st January 2018
What might happen if you don’t equilibrate your flash cartridges.
"To equilibrate or not to equilibrate" flash Silica cartridges prior to loading the crude material and what possible risks you might face if you skip this step, read here →
24th January 2018
“Alien vs. Predator”… or in other words “Silica vs. Alumina”
The rivalry between Silica and Alumina looks a lot like "Alien vs. Predator" movie, don't you think? Read more about how we can benefit from their "competition" →
17th January 2018
The “Golden Mean” in Flash Chromatography
Which system describes perfectly the balance ratio between productivity and cost in flash chromatography? Open columns, automated or semi-automated systems?








































































































































Peak tailing not achived i changed lot of columns Escitalopram oxalate please suggest what was the issue column or system or system software
Dear Meena,
Apologies for the delay in my response.
The primary cause of peak tailing is the occurrence of more than one mechanism of analyte retention.
For example: In reversed-phase (RP) separations, compounds possessing amine and other basic functional groups interact strongly with such silanol groups as in RP, producing tailing peaks.
There are a few methods that can be used to avoid peak tailing:
• Operate at a lower pH, use a buffer as phosphate butter (pH 4)
• Use a highly deactivated column (an ‘end-capped column’ as our PrepPure C18AQ columns)
• Consider the possibility of mass overload
• Consider the possibility of column bed deformation
I hope this helps, if not, please do not hesitate to leave another comment with more specific details regarding your application.
Kind regards,
Bart